
These rays represent the loss in energy when the remaining nucleons undergo stable rearrangements, and thus, gamma rays accompany other radioactive emissions. Gamma rays are electromagnetic radiations of short wavelengths and have no charge or mass. Instead, it involves photons of energy being emitted from an unstable radioactive nucleus. Gamma Radiation: Gamma radiation (γ) does not consist of any particles. In beta minus decay, the nucleus emits a neutron that is transformed into a proton (antineutrino) and an electron.īeta minus decay: 1 n → 1 p + + 0 -1 β - + v̅īeta plus decay: 1 1 p + → 1 0 n + 0 1 β + v In beta plus decay, the nucleus emits a positively charged electron (positron) and a proton that is converted into a neutron (neutrino). Beta-decay is of two types –beta plus and beta minus. Example: Uranium-238 undergoes alpha decay in the following manner:īeta Radiation: Consists of a stream of high-speed electrons. Thus, the resulting element has an atomic number less by two units and an atomic mass less by four units than that of the originating element. An alpha particle consists of two protons and two neutrons and is similar to a Helium-4 atom. For example: during Uranium-235 fission, the nuclear radiation that is emitted contains gamma-ray photons and neutrons.Īlpha Radiation: Alpha radiation is the emission of alpha particles when an atom goes through radioactive decay. Nuclear reactions release ionizing subatomic particles, including alpha particles, neutrons, beta particles, mesons, muons, positrons, and cosmic rays. Nuclear radiations include alpha rays, beta rays, and gamma rays. For this reason, nuclear radiation is also known as ionizing radiation. 
The particles emitted in nuclear reactions possess an energy that is tremendous enough to knock electrons from atoms and molecules, thereby ionizing them. Nuclear radiation refers to the photons and particles that are emitted during nuclear reactions. Nuclear chemistry is also termed radiochemistry, which involves the study of the elements composing the universe, design, and development of radioactive drugs for medicinal uses, and several other scientific applications. These nuclear changes are a source of nuclear power and radioactivity, and the energy released from the nuclear reactions has far-reaching applications. Nuclear chemistry is a sub-discipline of chemistry dealing with the study of changes in the nucleus of atoms of elements. This process is what she called radioactivity. Using an electrometer that could measure the electrical conductivity of air (the predecessor of the Geiger counter) invented by her husband Pierre and his younger brother Jack, she was able to prove that thorium also produces these rays. Marie Curie, who was the founder of nuclear chemistry, was intrigued by Antoine Henri Bekrel's discovery that photographic film can emit light that can be exposed even when uranium minerals are wrapped in black paper. You are also responsible for the maintenance of sample preparation materials and equipment, and for the safe use and disposal of samples and other materials used in the laboratory. You often work in the laboratory and may be responsible for the operation, maintenance, and repair of state-of-the-art equipment. Nuclear chemists carry out basic research, applied research, or theoretical research. They often work to improve the efficiency and safety of nuclear energy sources and the way radioactive materials are stored and disposed of. 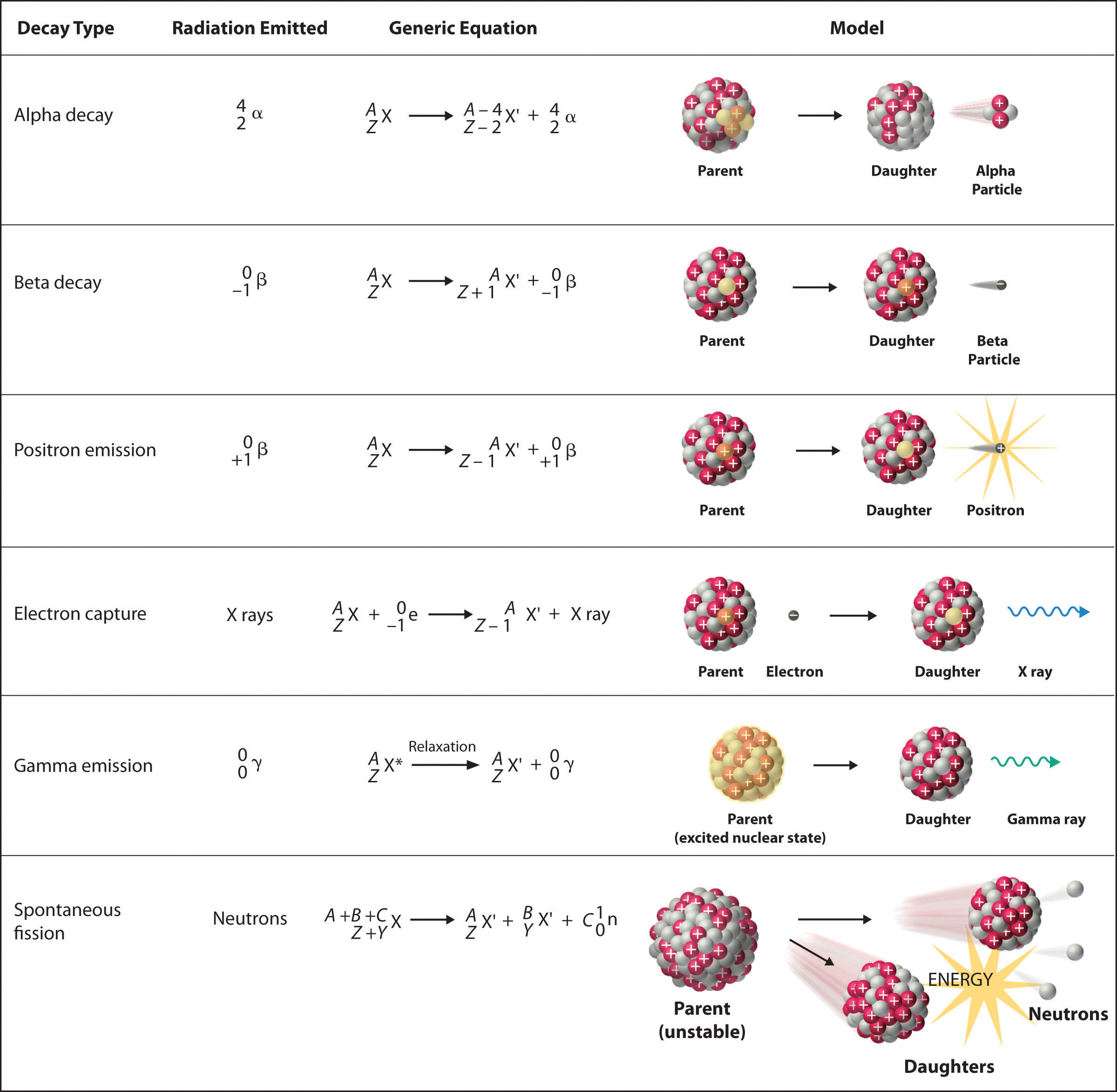
Nuclear chemists can be found in a variety of research areas, including nuclear imaging and nuclear technology. In fact, the chemical technology developed by nuclear chemists has become so important that biologists, geologists, and physicists use nuclear chemistry as a common tool in their disciplines. Modern nuclear chemistry sometimes referred to as radiochemistry, has become highly interdisciplinary in its applications, from the study of element formation in space to the design of radiopharmaceuticals for diagnostic medicine. 
Nuclear chemistry is the study of the chemical and physical properties of elements that deal with nuclear reactions or reactions that happen inside the structure of the nucleus. The wide proliferation of the area has brought about a wide variety of nuclear chemistry topics - what is nuclear chemistry, nuclear radiations, artificially simulated nuclear reactions (fission and fusion), and the uses of nuclear chemistry. From nuclear power generation to war damage, nuclear chemistry has shown tremendous potential. In the mid-twentieth century, novel findings and the Second World War ushered in the Nuclear Age. The discovery of radioactivity opened up the way for the creation and development of nuclear chemistry in the early twentieth century.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
